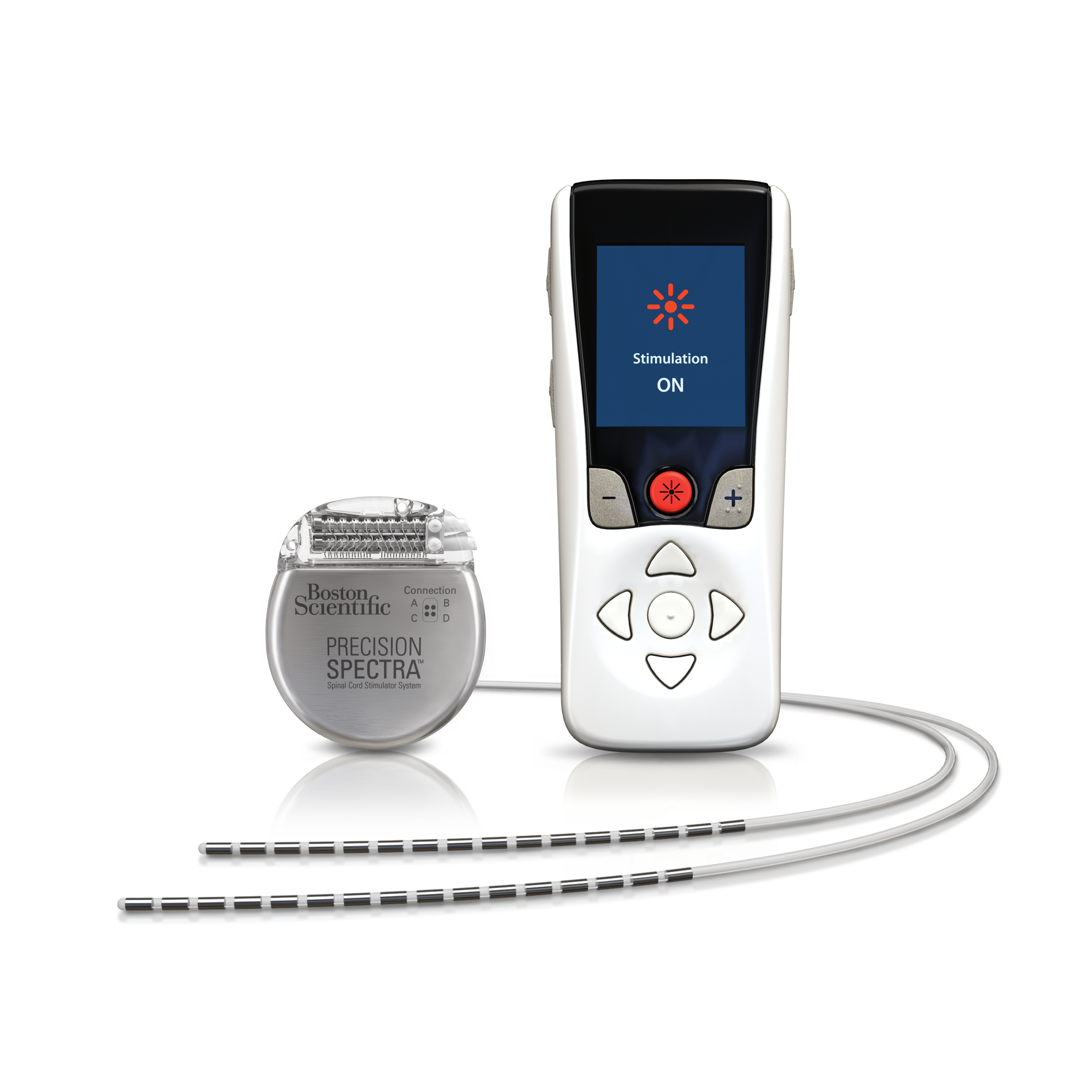
For therapy that does not generate paresthesia (i.e. Stimulation must be turned off first in such cases.
Patients using therapy that generates paresthesia should not operate motorized vehicles such as automobiles or potentially dangerous machinery and equipment with the stimulation on. Advise your physician that you have a Spinal Cord Stimulator before going through with other implantable device therapies so that medical decisions can be made and appropriate safety measures taken. The Spinal Cord Stimulator system may interfere with the operation of implanted sensing stimulators such as pacemakers or implanted cardiac defibrillators. The system should not be charged while sleeping. Strong electromagnetic fields, such as power generators or theft detection systems, can potentially turn the stimulator off, or cause uncomfortable jolting stimulation. As a Spinal Cord Stimulation patient, you should not have diathermy as either a treatment for a medical condition or as part of a surgical procedure. Exposure to MRI may result in dislodgement of the stimulator or leads, heating of the stimulator, severe damage to the stimulator electronics and an uncomfortable or jolting sensation.

Warnings. Patients implanted with Boston Scientific Spinal Cord Stimulator Systems without ImageReady™ MRI Technology should not be exposed to Magnetic Resonance Imaging (MRI). The Precision Montage™ MRI, WaveWriter Alpha™ and WaveWriter Alpha™ Prime SCS Systems with ImageReady™ MRI Full Body Technology are “MR Conditional” only when exposed to the MRI environment under the specific conditions defined in the applicable ImageReady™ MRI Full Body Guidelines for Precision Montage™ MRI or WaveWriter Alpha™ and WaveWriter Alpha™ Prime Spinal Cord Stimulator Systems. Patients implanted with the Precision Spectra™ or Spectra WaveWriter™ Spinal Cord Stimulator Systems with ImageReady™ MRI Technology are "MR Conditional" only when exposed to the MRI environment under the specific conditions defined in the applicable ImageReady™ MRI Head Only Guidelines for Precision Spectra™ or Spectra WaveWriter™ Spinal Cord Stimulator Systems.īoston Scientific’s ImageReady™ MRI Full Body Technology makes safe MRI scans possible. The Boston Scientific Spectra WaveWriter™, WaveWriter Alpha™ and WaveWriter Alpha™ Prime SCS Systems are also indicated as an aid in the management of chronic intractable unilateral or bilateral low back and leg pain without prior back surgery.Ĭontraindications. The Spinal Cord Stimulator systems are not for patients who are unable to operate the system, have failed trial stimulation by failing to receive effective pain relief, are poor surgical candidates, or are pregnant.īoston Scientific’s ImageReady™ MRI Technology makes safe MRI head scans possible. Indications for Use: The Boston Scientific Spinal Cord Stimulator Systems are indicated as an aid in the management of chronic intractable pain of the trunk and/or limbs including unilateral or bilateral pain associated with the following: failed back surgery syndrome, Complex Regional Pain Syndrome (CRPS) Types I and II, intractable low back pain and leg pain, Diabetic Peripheral Neuropathy of the lower extremities, radicular pain syndrome, radiculopathies resulting in pain secondary to failed back syndrome or herniated disc, epidural fibrosis, degenerative disc disease (herniated disc pain refractory to conservative and surgical interventions), arachnoiditis, multiple back surgeries.

